How Much Do Medical Devices Cost? Doctors Have No Idea; Some device makers don’t allow hospitals to disclose what they paid, driving up spending by insurers and Medicare
January 25, 2014 Leave a comment
How Much Do Medical Devices Cost? Doctors Have No Idea
By John Tozzi January 23, 2014
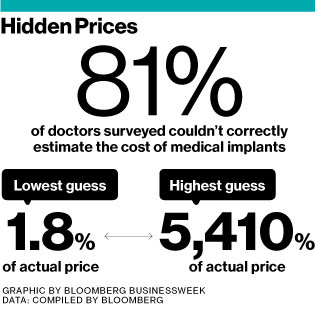
Imagine taking your car to a mechanic who has no clue how much a battery or muffler costs—and has no way of finding out. Substitute “artificial hip” for “battery” and “doctor” for “mechanic” and you get a pretty good picture of the convoluted market for medical implants. Asked to estimate the cost of common devices such as replacement knees or spinal screws, physicians at seven major academic hospitals in the U.S. were wrong 81 percent of the time, according to a January study published in the journalHealth Affairs. The survey of 503 orthopedists at institutions including Harvard, Stanford, and the Mayo Clinic considered doctors’ answers correct if they came within 20 percent of what their hospital paid suppliers. The worst guesses ranged from a small fraction of the actual price to more than 50 times what the hospital paid.
The doctors did so poorly in part because many medical device manufacturers require hospital purchasing departments to keep prices confidential, allowing sellers to charge some institutions more than others for the same products. “Widespread dissemination of device prices is not an option at many institutions,” note the authors of the study, which didn’t disclose what hospitals included in the survey paid. Prices “often varied considerably across institutions.”
Total spending on medical devices in the U.S. reached about $150 billion in 2010, or roughly a nickel of every health-care dollar, according to the Advanced Medical Technology Association (AdvaMed), the industry’s trade group. The device is often the most expensive part of an orthopedic procedure, and the bill is ultimately paid by either private insurers or Medicare and Medicaid. On average, hospitals paid suppliers $5,842 for an artificial hip in 2012, according to Orthopedic Network News; a replacement knee averaged $5,104. Stan Mendenhall, the publication’s editor, says that depending on their ability to negotiate with manufacturers, hospitals can pay anywhere from $2,000 to $16,000 for artificial hips and knees. A 2012 study by the U.S. Government Accountability Office also found wide variation in prices. “Some hospitals have substantially less bargaining power with the small group of companies that manufacture particular [implantable medical devices] and consequently face challenges in obtaining more favorable prices,” the GAO wrote.
Martin Makary, a surgeon at Johns Hopkins Hospital (which was not part of the Health Affairs survey), points to the example of surgical mesh, which depending on the type ranges in price from $75 to $10,000. Because doctors never see the insurance bill and often aren’t privy to how much various options cost, they have little incentive to choose a less expensive mesh from one company that may work as well as a costlier one from another. “We don’t know what the patients end up getting charged for it and if there is an up-charge,” Makary wrote in an e-mail.
A 2007 Senate bill would have required device makers to report their average prices to regulators and the public, but medical device makers successfully lobbied against it. “It’s a market that’s working extraordinarily well,” says David Nexon, AdvaMed’s senior executive vice president. He points to research, funded by the lobbying group, showing that spending on medical devices has remained at about 6 percent of total U.S. health-care costs since the early 1990s and that prices for major implants such as knees and hips have fallen since 2007.
In 2009 the Cleveland Clinic began using its clout to buck the system, enlisting surgeons on its staff to help contain costs by sharing prices with them and limiting the menu of devices they can choose from. Spinal implants that once came from 10 different companies have been narrowed to two, says Simrit Sandhu, who’s in charge of the clinic’s supply chain. Cleveland has kept some contracts with suppliers of expensive devices, but surgeons must justify using them instead of more economical options. Over the past four years, Sandhu says, the program has saved the hospital system $190 million. Last year, Cleveland Clinic formed a company called Excelerate Strategic Health Sourcing to help other hospitals copy its system.
Getting physicians to embrace such changes is easier in hospitals where doctors are salaried employees. John O’Brien, former president of UMass Memorial Health Care, says the Massachusetts hospital system encouraged its doctors to choose devices from a short list of suppliers, enabling the hospital to negotiate better prices.
That’s a harder sell with independent surgeons, who pick where they want to perform operations and can avoid hospitals that try to limit which devices they can use. “They don’t particularly care about the cost. They don’t know what the cost is,” says O’Brien, now a professor at Clark University in Worcester, Mass. For that reason, hospitals competing to attract surgeons to perform joint replacements and other lucrative procedures will continue stocking their preferred devices, even if less expensive ones would suffice. As O’Brien puts it, “You don’t want them to go across town.”
The bottom line: Some device makers don’t allow hospitals to disclose what they paid, driving up spending by insurers and Medicare.