Hepatitis C: Treated – at a price; New drugs that could potentially eliminate the disease are a boost for the industry
January 27, 2014 8:23 pm
Hepatitis C: Treated – at a price
By Andrew Jack
New drugs that could potentially eliminate the disease are a boost for the industry
Gemma Peppe lived with hepatitis C for two decades before she – and the teenage son she inadvertently infected at birth – received a first round of painful and ineffective treatment designed to stave off the risk of liver disease. “The side effects were dreadful,” she recalls. “You feel like you have flu, you become terribly angry and mentally unstable. My son stopped going to school and our house was like a bloodbath.”
Since 2012, both of their lives have been transformed after switching to an alternative experimental medicine that offered quicker, simpler and more effective treatment to them – and soon potentially to millions of other patients around the world.
Ms Peppe is working again in Britain and her son, after four years out of education, has resumed studying and won a university place to study mathematics. “It’s incredible. I thought I would die before my pension but now I feel like I’ve shed 15 years,” she says.
The launch in recent weeks of sofosbuvir
, the first of a series of new drugs for hepatitis C virus (HCV) set for regulatory approval, raises the prospect of curing and potentially eliminating a disease that levies a heavy global burden.
The new medicines will also provide a substantial financial boost to pharmaceutical companies after a fallow period in which there have been few significant “blockbuster” drugs, such as the cholesterol-lowering statins of the 1990s. Analysts say HCV medicines will generate billions of dollars in annual revenues, making them among the most lucrative of a new crop of drugs.
“This is a watershed moment for hepatitis C with newly available, highly effective, easy to administer therapies,” says Prof David Goldberg, an epidemiologist at Health Protection Scotland.
Yet in one of many parallels between HCV and HIV, sofosbuvir has sparked controversy over its high price. Gilead, the California company that produces sofosbuvir, known by the brand name Sovaldi, is charging $84,000 for the 12-week course of treatment in the US. Many patients will struggle to afford it in its home market. In poorer countries, still fewer are likely to gain access.
Hepatitis C in figures
● About 150m people are infected with the virus
● More than 350,000 people die each year from hepatitis C-related liver diseases
● The body fights off the virus in about 25 per cent of cases
● The market for hepatitis C treatments is forecast to hit$15.5bn by 2022
As Médecins Sans Frontières, the medical charity, argued before a discussion on the topic at the World Health Organisation: “New oral treatments set to become available in coming months will revolutionise hepatitis C treatment . . . However, for government programmes to be able to scale up and address the true burden of the epidemic, [they] must be affordable.”
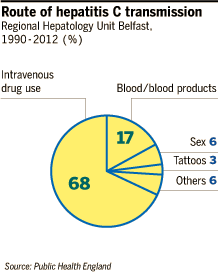
HCV infects an estimated 170m people globally. Like HIV, it can be transmitted sexually, via blood transfusion, from syringes shared by drug users, and from mothers to their children.
HCV appears more resilient, however, surviving more easily on inadequately sterilised surgical instruments and tattooing equipment. The UK National Health Service says it may be transferred via shared toothbrushes, razors or rolled banknotes used by those snorting cocaine.
There is no vaccine to prevent either HIV or HCV, and both typically take a number of years before symptoms develop. They are often long left undiagnosed, rendering eventual treatment more difficult and costly and increasing the risk of their inadvertent spread to others.
Without treatment HIV leaves the body at risk from opportunistic infections and almost inevitably death. By contrast, the body fights off HCV in about 25 per cent of cases. In the UK and other countries, HCV now kills more people than HIV.
Over 20 years or more, HCV causes liver disease. It is ultimately responsible for the majority of cirrhosis and liver cancer cases. Many of those infected may unwittingly consume large volumes of alcohol, intensifying and accelerating the effects of the disease on the liver.
Treatment is arduous. Patients require a year of regular injections of interferon and swallowing the pill ribavirin, which cause severe side effects. At best, only half of patients who complete their therapy are cured.

“The treatments are quite toxic,” says Barry Bernstein, head of antiviral development programmes at AbbVie, another drug company gearing up to launch new treatments. “Many patients decline them because they are so burdensome. People having injections on Friday night may spend the weekend in bed with flu-like symptoms and cannot then work.”
That explains the excitement surrounding the next generation of treatments. The parallels between the two viruses mean that the billions of dollars invested by universities and companies in developing HIV drugs over the past 30 years have helped understanding of how to tackle HCV.
Many of the researchers, such as Mr Bernstein, have worked on both diseases. So have more than a dozen drug companies: alongside AbbVie and Gilead, other large pharmaceutical producers in the field include Bristol-Myers Squibb, Merck, Roche,Johnson & Johnson and GlaxoSmithKline.
Some of the drugs they are studying – such as protease inhibitors designed to stop the viruses replicating – are closely related chemically to HIV equivalents. Researchers treating HIV discovered the need to attack the virus simultaneously with different types of medicines, ideally combined in a single pill. They are now applying the same principle to HCV.
“There were corollaries from HIV,” says John McHutchison, senior vice-president for liver disease therapeutics at Gilead. “The viruses are very similar, with a very high turnover and rate of replication. There is a highly reproducible cell culture system to test for molecules.”
Clinical trials and regulatory scrutiny are still required before many of the HCV drugs win approval, and their use without the need for interferon and ribavirin becomes possible. Not all are equally effective against different strains, nor for patients with other medical complications.
But the positive results in patients such as Ms Peppe and her son (who used AbbVie’s experimental compound) have increased interest.
They have also helped spur investments by drug companies.
“There’s a lot of hope associated with these drugs,” says Ueli Fankhauser, head of global product strategy at Roche. “The race is coming to the market and will result in tremendous benefit to the patient with the chances of being cured at 90-95 per cent.”
Vertex, the first company with an important recent breakthrough in hepatitis C, saw a surge in its valuation with the launch of Incivek in 2011. It generated sales of $1.6bn within its first 12 months on the market. A still stronger signal of the scientific and commercial potential of new treatments came when Gilead surprised the markets by paying $11.2bn for Pharmasset in 2012. That gave it control of the then still experimental drug Sovaldi.
In a research note last week, Barclays Capital forecast that Gilead would generate $416m in the first quarter this year alone from sales of the drug. “The highly anticipated and closely watched launch of Sovaldi has so far performed up to, if not above, expectations,” it said.
Others forecast that Sovaldi will overtake Incivek as the most rapid drug launch in the industry, generating $2.5bn this year. EvaluatePharma, a consultancy, judged the drug to be “easily the industry’s most valuable pipeline product”, estimating its annual sales would reach $7.4bn by 2018. Datamonitor, a market research specialist, says the market for all HCV treatments will total $15.5bn by 2022.
. . .
Yet Gilead’s strategy has also sparked criticism over the speed of progress and the affordability of its treatments – critiques likely to develop for its peers, too. It reflects another parallel between HCV with HIV: patient activists.
For many years, HIV patients were in the vanguard of medical campaigning, using sit-ins, demonstrations and political lobbying to accelerate funding, research and availability of treatment. HCV has never had a high political profile.
But many, including those with both viruses, are now reapplying their energies from HIV to its cousin.
Critics argue that Gilead delayed the launch of life-changing combination HCV therapy by refusing to test Sovaldi in combination with Bristol-Myers Squibb’s compound daclatasvir, despite results showing that the two drugs together generate high cure rates. Instead, Gilead combined Sovaldi with ledipasvir, its own experimental combination treatment, which had been further behind in the race to win regulatory approval.
The company has also priced Sovaldi expensively, arguing that its speed and efficacy justifies a premium over existing HCV therapies – not to mention the need to recover its development costs and the $11bn purchase price of Pharmasset.
Andrew Hill, a researcher at Liverpool University, argues that the new HCV drugs could be manufactured for just $100 to $200 for a course of treatment, and that high volume sales would offset lower prices.
“The question is whether you treat a very small number with low volumes and high margin, and have the epidemic carrying on for decades; or you treat many more with the chance to eradicate the disease,” he says.

“Given the overall size of the epidemic, even if prices were cut tenfold, the companies would still make a profit,” he says. He points out that in the UK alone, treating everyone with the disease at the current pricetag would cost £13bn, more than the annual NHS bill for all drugs.
Successful activism to extend access to antiretroviral drugs for HIV to poorer countries has encouraged advocates to do the same for HCV. Indian groups launched a legal challenge to Gilead’s Sovaldi patent application, seeking to allow generic companies to make it at lower cost.
“Once people see you can make a real difference with oral therapies, it spreads through the community very fast,” says Leena Menghaney of MSF. “The movement is being led by people who are not scared of demonstrating or going to jail. They are willing to do everything to get treatment.”
John Milligan, Gilead’s chief operating officer, points out that Sovaldi offers time and cost savings over existing HCV treatments – particularly when taking into account the savings from reduced liver disease.
But he also pledges to examine ways to make the drug available more cheaply in lower income countries, as Gilead did for its widely used HIV treatments. Unlike the latter disease, he points out that there are no donors to help support the costs.
HCV shares another characteristic with HIV: the need for prevention. The number of new infections remains high because of inadequate diagnoses, as well as poor blood and infection control standards.
With the new generation of drugs now becoming available, the pressures on governments to buy them – and companies to make them available affordably – are set to grow.
Charles Gore, head of the World Hepatitis Alliance, an umbrella organisation of patient groups, cautions: “It’s HIV all over again. Can we not make the same mistakes but instead learn from painful lessons? Or will companies wait for activists to set fire to their stands at scientific conferences?”
——————————————-
The good deed that led to disaster in Egypt
Alan Fenwick went to work in Egypt in 1988 to help tackle bilharzia, a parasitic worm disease that can cause severe liver damage. He discovered a country struggling with the legacy of past efforts to address the first problem, which had in turn made the burden of the second much heavier.
Today, the result is that Egypt has a far higher prevalence of hepatitis C than anywhere in the world: an estimated 15 per cent of the population aged 15-59 years old has been infected with the virus, imposing enormous costs on the health system. Nearly 200,000 people have HCV.
The problem began during the 1950s when a programme was launched to treat and prevent schistosomiasis (bilharzia), which infected people exposed to the Nile’s stagnant floodwaters. The good intentions had dreadful consequences.
The approach was to provide “mass drug administration” of tartar emetic, a poison injected into humans to kill the worms or prevent them spreading around the body. To do so, health workers unaware of the risks of infection used the same poorly sterilised glass syringes on multiple patients.
“The drug had to be given daily for 14 consecutive days,” recalls Prof Fenwick. “That’s an awful lot of cross needle work. They didn’t even know hepatitis C existed at the time, but when one person had it, they spread it.”
It was only in the 1990s that blood tests were developed for the virus and identified what Tom Strickland, a researcher at Imperial College in London, has described as Egypt’s “occult” hepatitis C epidemic. By then it had far overtaken bilharzia as the main cause of liver disease. The two conditions together make treatment still more complex.
The old injectable treatment has now long been replaced with the far safer pill praziquantel, in a control programme for bilharzia still overseen by experts including Prof Fenwick. Yet the underlying infection of hepatitis C has continued to rise and illness and death is projected to double over the next 20 years.
While such mass injection programmes no longer exist, Egypt still has low-quality infection control, permitting transmission through blood products, surgical and dental operations. Anecdotally, there are also examples of children scavenging hospital waste dumps for discarded syringes, which they sell for reuse.



