Unmeltable, Uncrushable: The Holy Grail in Painkillers
May 6, 2013 Leave a comment
Updated May 5, 2013, 9:18 p.m. ET
Unmeltable, Uncrushable: The Holy Grail in Painkillers
By TIMOTHY W. MARTIN and JONATHAN D. ROCKOFF
The arms race to build a safer painkiller is under way.
In the wake of a key decision by the Food and Drug Administration, more than a dozen pharmaceutical companies—from startups to Pfizer Inc. PFE -1.06% —are vying to create painkilling drugs that are difficult to abuse.
The FDA moved last month to block the manufacture and sale of generic versions of the original OxyContin, which has gone off patent but which is easier to abuse than the current version. A newer version of OxyContin, introduced by its maker, Purdue Pharma LP, in 2010, contains an infusion of polymer that makes the pill difficult to crush, meaning addicts can’t get all of the extended-release ingredients at once to get high.This week, the FDA must consider the effectiveness of another safeguarded, brand-name painkiller, Endo Pharmaceuticals Inc.’s ENDP +1.71% Opana ER. If the FDA thinks Opana ER slows abuse, the agency can shield Endo from the competition—since the generics don’t have any abuse-deterrents.
Such a decision would send a strong message to pharmaceutical makers that pain drugs with no safeguards are likely to be removed from the market—and that there are billions of dollars in potential revenue to be had from the sale of painkillers that are abuse proof.
As a result, companies such as Pfizer,Johnson & Johnson JNJ +0.69% and Endo are trying to crack that potentially lucrative market. Pfizer wants to introduce Remoxy, a direct OxyContin competitor with abuse deterrents, and a morphine drug called Embeda.
Johnson & Johnson is conducting tests to demonstrate abuse-deterrent properties of its opioid for moderate to severe pain. Purdue Pharma wants to adapt its abuse-deterrent technology for a hydrocodone product—a class of drugs currently dominated by low-cost generics that don’t have abuse-proof formulations.
“Over time, it should be a scientific race across the whole pharmaceutical industry to create a market where all opioids have abuse-deterrent properties,” said Gary L. Stiles, senior vice president of research and development at the closely held Purdue Pharma.
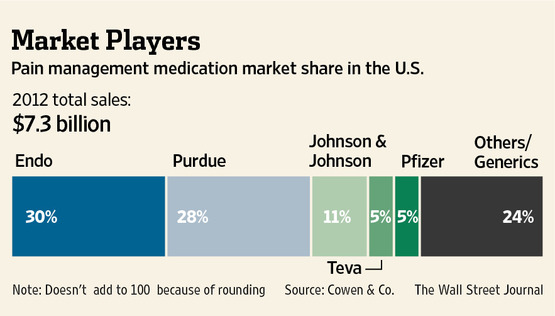
The race could give a boost to an already lucrative market and raise the cost of pain treatment. A recent Cowen & Co. report predicts an increase in prescription pain drug sales of more than 15% by 2017, reaching $8.4 billion, thanks in part to the FDA ruling. It could also bring about a reshuffling of market share in the fast-growing $7.3 billion pain market, as companies such as Pfizer move with full force in the opioid painkiller market for the first time.
The FDA isn’t directly requiring all painkillers to have safeguards. But should an effective abuse-deterrent drug be developed, the FDA said it has the power to remove on-the-market rival products which don’t.
Drug companies are racing to conduct the studies and research required by the FDA to prove their drugs thwart abuse. Winning that distinction from the FDA, as Purdue Pharma has done for OxyContin, allows company sales representatives to tout those properties to doctors—forming a potential new marketing battleground for branded drugs. “That’s how you’re going to see the battle going forward,” said Mike Royal, chief of clinical affairs at AcelRx Pharmaceuticals Inc., ACRX -0.69% which is developing treatments for acute pain.
Pfizer could grab more of the prescription painkiller market, based on its product pipeline and ability to successfully market new drugs, analysts said. Endo currently leads the pain management drugs market with a 30% share, followed by Purdue Pharma’s 28% share, according to a recent Cowen & Co. report. Pfizer had just 5%, according to Cowen.
Pfizer has been lying in wait. The company’s $3.6 billion acquisition of King Pharmaceuticals in 2011 took it into the opioid market. In addition to Remoxy and Embeda, Pfizer owns the rights to at least five other painkillers.
Remoxy is protected from abuse by a type of sugar sucrose that is used in orange soda or energy drinks. It prevents the medication from splitting apart or separating under the duress of heat or being mixed with alcohol, said James E. Brown, chief executive of Durect Corp., which developed Remoxy with Pain Therapeutics Inc.PTIE +4.16% before selling it to King.
The FDA rejected approval of Remoxy in 2011, but Pfizer met with the FDA in March and is discussing the “next steps” to address the issues raised, said John Young, Pfizer’s president of primary-care business, on a Tuesday earnings call with analysts. “We believe we have a path forward,” he said, adding Embeda could launch in the first half of 2014.
The FDA is expected to decide Friday whether Endo’s Opana, which turns into a jellylike substance when abusers heat it up for injection, can be marketed to physicians as having abuse-deterrent features.
Johnson & Johnson is also conducting studies to show the abuse-deterrent properties of Nucynta ER and plans on meeting with the FDA later this year to review it, said Gary Vorsanger, senior director of clinical development at the company’s Janssen Pharmaceuticals unit.
Purdue Pharma, for its part, says it is looking for ways to use the OxyContin technology on other drugs, like other formulations of oxycodone, morphine and hydrocodone.
Smaller biotech firms are crafting their own safeguards. Egalet Ltd., of Malvern, Pa., has turned to a plastic injection molding manufacturing technique used to make Legos or plastic bottle caps. The pills are so hard that when placed in a coffee grinder—a common way to break down pills by abusers—the blades get chipped. “It’s a very physically imposing pill,” said Bob Radie, Egalet’s chief executive.
North Liberty, Iowa-based KemPharm Inc. is developing a hydrocodone product bonded with a common additive—benzoic acid—that’s used to prevent growth of mold and other bacteria in foods like pickles, yogurt and beer. Addicts trying to inject or inhale the pills won’t feel a thing, because the medication can only be broken down by powerful enzymes in the stomach or intestinal track.
“What you get, essentially, is nothing. It’s no better than ordinary table salt or sugar,” said KemPharm Chief Executive Travis Mickle.